We'll be back in a bit !
The system is currently undergoing a routine upgrade to ensure you get the best learning experience. The maintenance is expected to continue till 8:00 pm. Please check back later.
Thank you for your understanding!
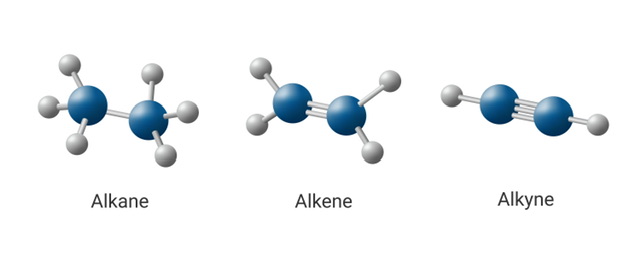
Alkane, alkene, and alkyne are kinds of aliphatic hydrocarbons. Aliphatic hydrocarbons are carbon- and hydrogen-based organic molecules. They may be found in many different forms, from natural gas and gasoline to polymers, and are the most common organic substances on Earth. There are two primary classes of aliphatic hydrocarbons, saturated and unsaturated. We can't imagine our existence without aliphatic hydrocarbons. Several industries put them to use, from the energy sector to the plastics and pharmaceutical industries. Aliphatic hydrocarbons are ubiquitous in the things we use and in the natural world, so it's important to know what makes them tick.
Alkane, alkene and alkyne
The organic molecules known as alkanes contain simply carbon and hydrogen. These are the most common organic compounds and the simplest hydrocarbons. Characteristics of these include:
Saturated hydrocarbons, like alkanes, have only a single link between their carbon atoms. There is no localized area of partial positive or negative charge in alkanes, making them another type of non-polar molecule.
All three of these sources—crude gasoline, oil and gas, and coal—contain alkanes, the most prevalent kind of hydrocarbon. Furthermore, alkanes can be employed as fuels, lubricants, and solvents. According to their carbon-atom count, alkanes are given their respective names.
Methane, with only one carbon atom, is the simplest alkane. Ethane, which consists of two carbon atoms, is the next alkane in the sequence.
An alkane may be described by the formula CnH2n+2, where n is the total number of carbon atoms. Due to their lack of polar groups and strong carbon-carbon bonds, alkanes are not very reactive. They are relatively stable and do not readily react with other chemicals, making them suitable as fuels.
In contrast, alkanes may be oxidized to produce a variety of other compounds, including alcohols, aldehydes, and ketones. Due to their ability to dissolve both polar and nonpolar molecules, alkanes are also widely employed as solvents. They are also put to good use as lubricants due to their slippery nature and ability to lessen friction.
Organic molecules with a carbon-carbon double bond are known as alkenes. This is because they are unsaturated hydrocarbons, which have fewer hydrogen atoms than their alkane counterparts. Organic chemists value alkenes for their versatility in compound formation.
In the formation of alkenes, two carbon atoms share two pairs of electrons. This results in an unsaturated alkene by forming a double bond between the two carbons. In addition to the double bond's other functions, alkenes are highly reactive. It is possible to perform addition, elimination, and substitution processes on alkenes.
To perform an additional reaction, a reagent like hydrogen is added to an alkene's double bond. An alkane is produced as a result. The double bond of an alkene can undergo elimination reactions, which include the removal of a reagent like water. An alkene is produced as a consequence. The double bond of an alkene can undergo substitution processes, in which one or more atoms are exchanged for another.
The creation of polymers also requires alkenes. When the same building blocks of molecular structure are repeated, the resulting macromolecules are called polymers. As alkenes may be readily bonded with one another, they are frequently used in the production of polymers. Because of this, they may be used effectively in the manufacturing of various plastics, rubbers, and some other products.
The manufacture of fuels also relies heavily on alkenes. A variety of fuels, including gasoline, diesel, and others, may be synthesized from alkenes. Lubricants, solvents, as well as other chemical products are all made from alkenes.
The presence of a triple bond between any two carbon atoms defines a hydrocarbon as alkyne. When compared to the maximum amount of hydrogen atoms that may be attached to a certain number of atoms of carbon, alkynes have less. This makes them an unsaturated hydrocarbon. The existence of an acetylene group, which is made up of two carbon atoms and a triple bond, gives alkynes another name: acetylenes.
Reactivity makes alkynes useful in organic chemistry. Because of its reactivity, the triple bond may be exploited to create many different molecules. It is possible to convert alkynes into a variety of other compounds, such as alcohols, aldehydes, ketones, and carboxylic acids.
Pharmaceuticals, dyes, and several other industrial compounds all make use of alkynes in their manufacturing. Moreover, alkynes are employed as solvents in the paint and varnish-making process, and as additives in gasoline.
The number of carbons in an alkyne molecule is used to categorize it. Two carbons and a single triple bond make ethyne the simplest alkyne. Propyne (with three carbon atoms), butyne (with 4 carbon atoms), and pentyne are also prevalent alkynes (five carbon atoms).
IUPAC nomenclature is used to provide names to alkynes. With a prefix indicating the number of carbon atoms, the name of the matching alkane is modified by adding the suffix -yne. As an illustration, ethane is renamed ethyne with the -yne suffix.
As a result of their versatility and high reactivity, alkynes play a significant role in organic chemistry. They serve as additives to gasoline and as solvents among their many other uses. The International Union of Pure and Applied Chemistry (IUPAC) nomenclature system is used to provide names to alkynes and to categorize them based on the number of carbon atoms they contain.
Feature | Alkane | Alkene | Alkyne |
| General Formula | CnH2n+2 | CnH2n | CnH2n-2 |
| Type of Bond | Single bonds only | At least one double bond | At least one triple bond |
| Saturation | Saturated hydrocarbons | Unsaturated hydrocarbons | Unsaturated hydrocarbons |
| Examples | Methane (CH4), Ethane (C2H6) | Ethene (C2H4), Propene (C3H6) | Ethyne (C2H2), Propyne (C3H4) |
| Reactivity | Least reactive | More reactive than alkanes | Most reactive |
| Structure | Single C-C bonds, tetrahedral | Double bond causes a planar structure | Triple bond causes a linear structure |
| Combustion | Burns with a clean flame | Burns with a sootier flame | Burns with a sooty flame |
| Addition Reaction | Do not undergo addition | Undergo addition reactions | Undergo addition reactions |
In organic chemistry, three types of hydrocarbons—alkanes, alkenes, and alkynes—are often encountered. Saturated hydrocarbons, such as those found in alkanes, have every single one of their carbon atoms connected to the maximum amount of hydrogen atoms possible. As unsaturated hydrocarbons, alkenes have a double bond between two carbon atoms. Nevertheless, alkynes differ from other unsaturated hydrocarbons in that they include at least one carbon atom that is triple-bonded.
It is important to know concepts like Alkane, Alkene and Alkyne in building a strong foundation in science. At 88tuition, we offer PSLE Science tuition classes to help students understand these concepts easily. Also, we provide clear video lessons, simple explanations and guided practice to support learning. Our structured approach allows your child to develop a deep understanding of science. So, they can be well prepared for their PSLE exams.
Frequently Asked Questions
1. Which is the least reactive hydrocarbon amongst alkane, alkene and alkyne?
Due to their stability, alkanes are the hydrocarbons with the lowest reactivity. Only robust single sigma bonds are present in them. Sigma bonds are the least reactive since they take a lot of energy to be broken.
2. Do alkanes undergo ozonolysis?
Alkanes do not undergo ozonolysis. It is a characteristic reaction of alkenes because it is a sort of addition reaction which is followed by bond cleavage. Alkane reactions do not show additional reactions.
3. Why are some alkynes acidic?
Due to the increased electronegativity of sp- hybridized carbon, terminal alkynes exhibit acidic behavior (triple-bonded carbon). This trait, however, is absent in alkanes and alkenes. Alkyne's connected hydrogen, which is triple-bonded to carbon, can dissociate into H+ ions.
